Close
Sign in to take advantage of your best pricing.
Everyday Free Shipping on Qualifying Orders Over $69.
Learn more about our FREE Environments Design Services.
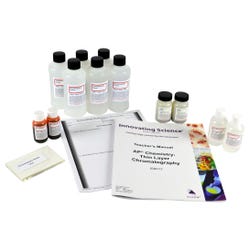
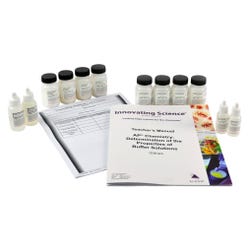
Advanced Placement
page_infoFilterskeyboard_arrow_down
Filters
Press enter to collapse or expand the menu. Clear AllFilters
Product List
[
{
"catentry_id" : "3074457345616694767",
"Attributes" : { }
}
]
[
{
"catentry_id" : "3074457345616697255",
"Attributes" : { }
}
]
[
{
"catentry_id" : "3074457345616697306",
"Attributes" : { }
}
]
[
{
"catentry_id" : "3074457345616699457",
"Attributes" : { }
}
]
[
{
"catentry_id" : "3074457345616699497",
"Attributes" : { }
}
]
[
{
"catentry_id" : "3074457345616699729",
"Attributes" : { }
}
]
[
{
"catentry_id" : "3074457345616701131",
"Attributes" : { }
}
]
[
{
"catentry_id" : "3074457345616763473",
"Attributes" : { }
}
]
[
{
"catentry_id" : "3074457345616764192",
"Attributes" : { }
}
]
[
{
"catentry_id" : "3074457345616765314",
"Attributes" : { }
}
]
[
{
"catentry_id" : "3074457345616771282",
"Attributes" : { }
}
]
[
{
"catentry_id" : "3074457345616772209",
"Attributes" : { }
}
]
[
{
"catentry_id" : "3074457345616782158",
"Attributes" : { }
}
]
[
{
"catentry_id" : "3074457345616808664",
"Attributes" : { }
}
]
[
{
"catentry_id" : "3074457345616814336",
"Attributes" : { }
}
]
[
{
"catentry_id" : "3074457345616815575",
"Attributes" : { }
}
]
[
{
"catentry_id" : "3074457345616819312",
"Attributes" : { }
}
]
[
{
"catentry_id" : "3074457345616822173",
"Attributes" : { }
}
]
[
{
"catentry_id" : "3074457345619506686",
"Attributes" : { }
}
]
[
{
"catentry_id" : "3074457345619506690",
"Attributes" : { }
}
]
[
{
"catentry_id" : "3074457345619506691",
"Attributes" : { }
}
]
[
{
"catentry_id" : "3074457345619506693",
"Attributes" : { }
}
]
[
{
"catentry_id" : "3074457345619506696",
"Attributes" : { }
}
]
[
{
"catentry_id" : "3074457345619506700",
"Attributes" : { }
}
]
[
{
"catentry_id" : "3074457345619506703",
"Attributes" : { }
}
]
[
{
"catentry_id" : "3074457345619506719",
"Attributes" : { }
}
]
[
{
"catentry_id" : "3074457345619506723",
"Attributes" : { }
}
]
[
{
"catentry_id" : "3074457345619506726",
"Attributes" : { }
}
]
[
{
"catentry_id" : "3074457345619506728",
"Attributes" : { }
}
]
[
{
"catentry_id" : "3074457345619506730",
"Attributes" : { }
}
]
[
{
"catentry_id" : "3074457345619506733",
"Attributes" : { }
}
]
[
{
"catentry_id" : "3074457345619506747",
"Attributes" : { }
}
]
[
{
"catentry_id" : "3074457345619506755",
"Attributes" : { }
}
]
[
{
"catentry_id" : "3074457345619506760",
"Attributes" : { }
}
]
[
{
"catentry_id" : "3074457345619506769",
"Attributes" : { }
}
]
[
{
"catentry_id" : "3074457345619506779",
"Attributes" : { }
}
]
[
{
"catentry_id" : "3074457345619506792",
"Attributes" : { }
}
]
{ searchResult: {
pageSize: 48,
searchTerms: '',
totalPageNumber: 1.0,
totalResultCount: 37,
currentPageNumber:1,
attributes: ""
}
}